WASHINGTON, D.C. (June 15, 2021)—The Department of Health and Human Services (HHS) announced important new information for recipients of the CARES Act Provider Relief Fund (PRF). Below are the key changes:
- HHS removed the deadline for all funds to be used by June 30, 2021. The deadline to use funds is now based on the date payments were deposited.
- Recipients can begin submitting information in the Reporting Portal starting July 1, 2021.
- Previously, HHS required recipients that received $10,000 in aggregate from the PRF to submit information in the Reporting Portal. Instead, HHS is now requiring recipients that received $10,000 or more in aggregate within a Payment Received Period to submit a report.
- Recipients will now have 90 days to complete the reporting requirement. Previously, recipients only had 30 days.
Deadline to Use Funds Now Based on Payment Received Period
Previously recipients were required to use all funds by June 30, 2021. The new FAQ states the use of funds deadline is now based on the time period the funds were deposited to the recipient. Below is a chart provided by HHS.
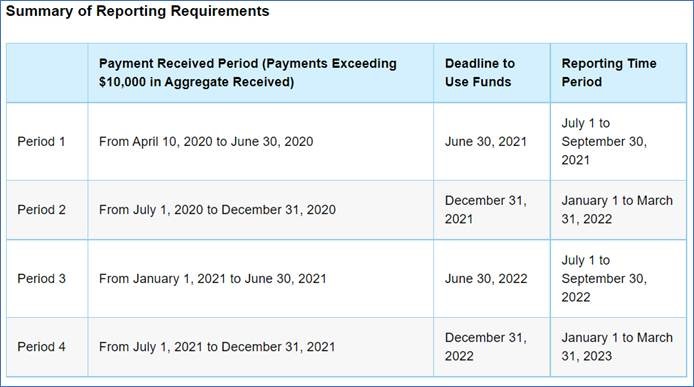
Any payment that is not used by the deadline towards acceptable expenses or losses will need to be returned to HHS.
HHS updated the Provider Relief Fund General and Targeted Distribution Post-Payment Notice of Reporting Requirements with additional information. The document includes information on steps to complete the report, data elements that will be collected, and more.
“These updated requirements reflect our focus on giving providers equitable amounts of time for use of these funds, maintaining effective safeguards for taxpayer dollars, and incorporating feedback from providers requesting more flexibility and clarity about PRF reporting,” said Diana Espinosa, acting administrator of the Health Resources and Services Administration (HRSA).
“The substantial relief for HME suppliers and other health care entities through the Provider Relief Fund has helped mitigate the rising product costs and new operational requirements stemming from the COVID-19 Public Health Emergency (PHE)," Tom Ryan, president and CEO of AAHomcare, said. "Unfortunately, many of these higher costs are likely to persist well beyond the end of the PHE thanks to continued supply chain constraints, basic material costs and potential inflation. HME suppliers deserve reimbursement rates that reflect these new market realities and will allow us to effectively serve the vulnerable patient groups that depend on us—that’s the kind of relief we really need going forward.”
“We are pleased that the Department of Health and Human Services is providing a limited extension on providers’ use of funds, and we are pleased to see some clarification on reporting,” said National Association for Home Care & Hospice president Bill Dombi. “We do hope HHS will consider further extensions on the use of funds due to the immense pressures on providers brought on by the public health emergency.”
The reporting requirements do not apply to COVID-19 coverage assistance funds, the Rural Health Clinic COVID-19 Testing Program, or the HRSA uninsured program.
Reportedly, about $24 billion of the $178 billion in the provider relief fund has not been distributed.
Visit the Provider Relief Fund website for more information and to view the updated FAQs.
