
When the Centers for Medicare & Medicaid (CMS) released its final rule (CMS-3819-F) last year, it not only revised and modernized the conditions of participation (CoPs) that home health agencies must meet in order to participate in programs, it presented new conditions. One such condition is §484.65, Quality Assessment and Performance Improvement (QAPI).
The effective date of the final HHA CoP was January 13, 2018, but agencies have until July 13, 2018 to implement at least one performance improvement project (PIP), which is part of the new QAPI condition.
HHAs must develop, implement, evaluate and maintain an effective, ongoing, HHA-wide, data-driven QAPI program. The HHA’s governing body must ensure the program reflects the complexity of its organization and services; involves all HHA services (including those services provided under contract or arrangement); focuses on indicators related to improved outcomes, including the use of emergent care services, hospital admissions and readmissions; and takes actions that address the HHA’s performance across the spectrum of care, including the prevention and reduction of medical errors.
CMS has not issued any standardized forms to meet this condition. Agencies are left to create their own forms and reporting tools. Many agencies are finding it difficult to know what to measure, how to measure, how to document their efforts and how to select a PIP. This article will help toward compliance.
QAPI is the merger of two complementary approaches to quality management, quality assurance (QA) and performance improvement (PI). QA is a reactive and retrospective while PI, is pro-active and continuous.
According to Shireesha Jevaji of the National Committee for Quality Assurance (NCQA), a key aspect of quality assurance is systematic measurement: monitoring processes, with a feedback loop for error prevention.
There are five standards in the QAPI requirements: 1) program scope, 2) program data, 3) program activities, 4) performance improvement projects (PIP), and 5) executive responsibility. The standards are not stand-alone or sequential. They flow together and cross over each other.
HHAs must also maintain and document an infection control program to prevent and control infections, along with communicable diseases. Infection prevention and control must be integrated into your QAPI policy.
1. Program Scope
§484.65(a)(1)The program must at least be capable of showing measurable improvement in indicators for which there is evidence that improvement in those indicators will improve health outcomes, patient safety and quality of care.
To meet this standard: Utilize a model for improvement including the Plan-Study-Do-Act (PSDA) cycle to help identify a target area that is measurable and will improve outcomes, patient safety or quality of care. Target areas that meet this criterion are consistent across the industry and include patient falls, hospital readmissions, emergency department visits and patient infections, developing or worsening pressure ulcers and medication errors, to name a few.
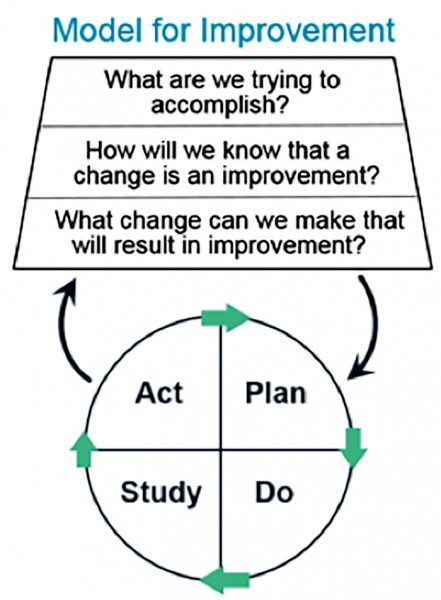 Plan-Do-Study-Act Cycle
Plan-Do-Study-Act Cycle§484.65(a)(2) The HHA must measure, analyze, and track quality indicators, including adverse patient events, and other aspects of performance that enable the HHA to assess processes of care, HHA services, and operations.
To meet this standard:
Report and analyze potentially avoidable events, publicly reported outcomes, OASIS, Home Health Compare, HHCAHPs, triage/on-call logs, competency assessments, chart audits, adverse drug events, infections, falls, patient complaints, clinical or operational processes and other reports as indicated. Access all available reports to identify potential areas of concerns. Ensure the agency has an organized tracking system for these reports and the trending of data. Each area reviewed (or indicator) must be measurable with the ability to track the effectiveness in any change in procedures, policies or interventions.
The QAPI program must include procedures for and frequency of measurement and analysis of indicators.
2. Program Data
§484.65(b)(1) The program must utilize quality indicator data, including measures derived from OASIS, where applicable, and other relevant data, in the design of its program.
§484.65(b)(2) The HHA must use the data collected to: §484.65(b)(2)(i) Monitor the effectiveness and safety of services and quality of care; and §484.65(b)(2)(ii) Identify opportunities for improvement.
§484.65(b)(3) The frequency and detail of the data collection must be approved by the HHA’s governing body.
To meet this standard: Utilize the data gathered and analyzed in the program scope standard. Identify opportunities for improvement. Focus on quality and safety issues. Measure and record quality indicators and areas that can stimulate performance improvement. Assign a focus group to review data and provide feedback. Establish benchmarks for comparisons. Prioritize and set goals. Remember: The governing body must be involved and ensure the agency systematically collects data to measure various aspects of quality of care; the frequency of data collection and how the data will be collected and analyzed (who, what, when and how often). Record these actions in the minutes of the governing body meetings. Keep a copy of the minutes in your QAPI binder.
3. Program Activities
§484.65(c)(1) The HHA’s performance improvement activities must:
§484.65(c)(1)(i) Focus on high risk, high volume or problem-prone areas; §484.65(c)(1)(ii) Consider incidence, prevalence and severity of problems in those areas; and §484.65(c)(1)(iii) Lead to an immediate correction of any identified problem that directly or potentially threatens the health and safety of patients.
§484.65(c)(2) Performance improvement activities must track adverse patient events, analyze their causes, and implement preventive actions.
The Potentially Avoidable Event Report in Casper displays incidence rates for 12 untoward events. Each of the Potentially Avoidable Events should be investigated and actions taken to reduce incidence. To determine if one of these measures would be appropriate for a PIP, compare your current data with national data and to your prior time period. The incidence rates have been adjusted to account for variation in patient characteristics. Those events with higher incidence and/or those that have the most clinical relevance to the agency and patient safety should be considered for a PIP.
§484.65(c)(3) The HHA must take actions aimed at performance improvement, and, after implementing those actions, the HHA must measure its success and track performance to ensure that improvements are sustained.
To meet this standard: Focus on high-risk, high-volume or problem-prone areas.
If a problem is identified which directly or potentially threatens the health and safety of a patient, correct the problem immediately.
Use Root Cause Analysis (RCA) to identify causative factors or root of the problem. Most of the accrediting bodies require homecare agencies to use the RCA process for investigating adverse or sentinel events. There are many free templates to guide an RCA.
Implement a corrective action plan focusing on the causative factors identified in your RCA. Continue to utilize the PSDA model.
- High risk: significant risk to the health or safety of patients
- High volume: frequently provided to a large patient population
- Problem-prone: potential for negative outcomes that are associated with a diagnosis or condition
- Adverse patient events: negative and unexpected impacting the plan of care and potential decline of the patient
4. Performance Improvement Projects
Beginning January 13, 2018, HHAs must conduct performance improvement projects.
§484.65(d)(1) The number and scope of distinct improvement projects conducted annually must reflect the scope, complexity and past performance of the HHA’s services and operations.
§484.65(d)(2) The HHA must document the quality improvement projects undertaken, the reasons for conducting these projects, and the measurable progress achieved on these projects.
To meet this standard: The HHA should have one performance improvement project either in development, ongoing or completed each calendar year. By July 13, 2018 at least one PIP must be in place.
The HHA decides, based on the QAPI program activities and data, what projects are indicated and the priority of the projects. Projects should be selected based on areas identified to be problematic, poor outcomes, high-risk or high-volume, and they must be individualized to your agency.
Document progress toward the PIP goals in measurable terms. Present updates to the HHA governing body at least quarterly and obtain feedback for modification of the PIP. Identify resources needed to put the plans into action. Tools should be utilized to measure and track progress of action items. Obtain measurements and data at a regular and consistent interval to validate progress or decline in goals.
Below is an example of tracking the action items in a PIP.
 Tracking PIP action items
Tracking PIP action items5. Executive Responsibilities
The HHA’s governing body is responsible for ensuring the following:
§484.65(e)(1) That an ongoing program for quality improvement and patient safety is defined, implemented, and maintained; §484.65(e)(2) That the HHA-wide quality assessment and performance improvement efforts address priorities for improved quality of care and patient safety, and that all improvement actions are evaluated for effectiveness; §484.65(e)(3) That clear expectations for patient safety are established, implemented and maintained; and §484.65(e)(4) That any findings of fraud or waste are appropriately addressed. Interpretive Guidelines §484.65(e)
To meet this standard: The agency’s governing body must assume responsibility for your QAPI program and ensure documentary evidence of the program. Keep minutes from all QAPI meetings.
Educate staff at all levels on the QAPI program and each individual’s role in the success of your QAPI program.
An HHA must maintain documentary evidence of its QAPI program and be able to demonstrate its operation to CMS.
In the event the HHA identifies a possibly illegal action by its employees, contractors or responsible/relevant physicians, it is the responsibility of the HHA to report the actions to the appropriate authorities according to the individual state laws and the nature of the action(s).
The QAPI program must be reviewed annually. The final rule (CMS-3819-F2) is available on the Federal Register website.
